Fetuin-A/alpha-2-Heremans Schmid's glycoprotein (FETUA/AHSG), Fetuin-B (FETUB), Histidine-rich glycoprotein (HRG) and Kininogen (KNG) are members of the cystatin superfamily, a subgroup of cysteine proteinase inhibitors. They form the so-called type 3 members, which are all synthesized in the liver and secreted into the blood. All type 3 cystatins have several disulfide bridges. Each protein is encoded by a single gene. The genes are located in close proximity in all known vertebrate genomes and are closely related. Figure 1 schematically shows the protein structure of type 3 cystatins.
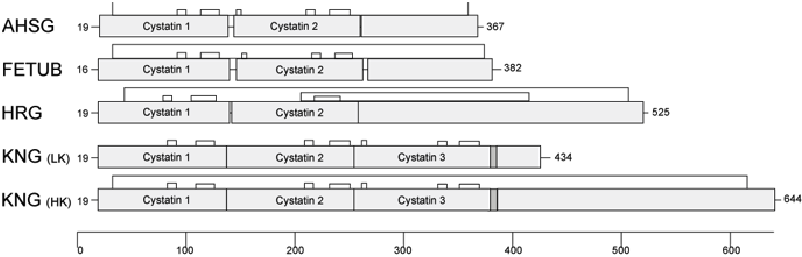
The fetuin genes were created by duplication of cystatin protein domains and fusion with other stable gene units. The combination of stable protein building blocks conserved during evolution produced proteins that are very similar in structure but have completely different functions. This can be best explained using the example of the proteins Fetuin-A and Fetuin-B. Both proteins have three domains, are similar in size, are made mainly in the liver and are secreted into the blood. Through genetic studies on mouse strains from which the respective genes were specifically removed, using so-called knockout mice, we found out that Fetuin-A is mainly responsible for the transport and stability of minerals in the body, whereas Fetuin-B is highly specific in inhibiting so-called zinc metalloproteinases. Despite the close relationship of the proteins, the knockout mice therefore show strikingly different phenotypes, i.e. the lack of Fetuin-A and Fetuin-B has completely different consequences. Fetuin-A knockout mice show calcium deposits throughout the body, which lead to restrictions in organ function with increasing age, whereas Fetuin-B knockout mice are completely infertile as females, because the shell of the eggs of these females harden before fertilisation and thus prevent fertilisation.
In addition to these clearly detectable functions, both proteins probably have other, subordinate functions that are emphasised elsewhere but which cannot be detected in the knockout mice. This does not mean that such functions cannot be observed experimentally, but in contrast to the functions just described, they cannot be consistently demonstrated by genetic, biochemical, molecular biological and clinical studies, but often only by individual observation on selected experimental systems.
Introductions to the function and mode of action of the fetuin proteins can be found in the following review articles and the literature cited therein, as well as in the list of literature which opens by clicking on the menu item "Publications". For further details see "Biomineralisation" and "Reproductive Biology" on the homepage. Enjoy reading!
Further Reading
Type 3 cystatins; fetuins, kininogen and histidine-rich glycoprotein.
Lee C, Bongcam-Rudloff E, Sollner C, Jahnen-Dechent W, Claesson-Welsh L.
Front Biosci (Landmark Ed). 2009 Jan 1;14:2911-22. Review.
PMID: 19273244
Nature's remedy to phosphate woes: calciprotein particles regulate systemic mineral metabolism.
Jahnen-Dechent W, Smith ER.
Kidney Int. 2020 Apr;97(4):648-651. doi: 10.1016/j.kint.2019.12.018. PMID: 32200857
Fetuin-A regulation of calcified matrix metabolism.
Jahnen-Dechent W, Heiss A, Schäfer C, Ketteler M.
Circ Res. 2011 Jun 10;108(12):1494-509. doi: 10.1161/CIRCRESAHA.110.234260. Review. PMID: 21659653
Biomineralisation and Calciprotein particles
The minerals calcium and phosphate are indispensable for the cellular metabolism of all living organisms. Calcium is both a cellular messenger and an essential component in bones and teeth. Energy-rich phosphates are also instrumental in energy metabolism and control the action of signaling molecules. Both ions typically occur in millimolar concentrations in biological fluids. This creates a solubility and transport problem; calcium phosphates are poorly soluble in water and readily precipitate from supersaturated solutions. A similar transport problem exists with fats, especially cholesterol, which must also be transported in large quantities in the aqueous environment of the blood, but is virtually insoluble in water. If fats and minerals are colloidally stabilized with the help of proteins, transport in the body can take place.
Blood thus contains dissolved components as well as colloidally stabilized substances in addition to blood cells. Calciprotein particles (CPP) are colloidal protein-mineral complexes discovered at our Institute. CPP are formed in protein-containing solutions supersaturated with calcium and phosphate. In a protein-free solution with an excess of calcium and phosphate, crystalline calcium phosphate precipitates immediately. Proteins in the solution bind small calcium phosphate clusters and prevent their further growth into crystals, stabilizing the solution. We study the formation, structure and function of calciprotein particles in bone mineralization and pathological calcification. We measure the calcification risk in patients with disturbed mineral balance.
Further Reading
Mud in the blood: the role of protein-mineral complexes and extracellular vesicles in biomineralisation and calcification.
Jahnen-Dechent W, Büscher A, Köppert S, Heiss A, Kuro-o M, Smith ER.
J Struct Biol. 2020;212:107577
Nature's remedy to phosphate woes: calciprotein particles regulate systemic mineral metabolism.
Jahnen-Dechent W, Smith ER.
Kidney Int. 2020 Apr;97(4):648-651.
Fetuin-A regulation of calcified matrix metabolism.
Jahnen-Dechent W, Heiss A, Schäfer C, Ketteler M.
Circ Res. 2011 Jun 10;108(12):1494-509.
Reproductive Biology
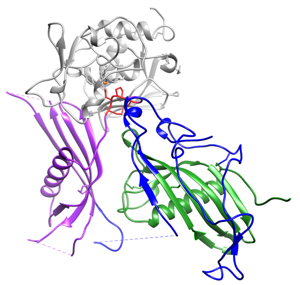
The plasma protein Fetuin-B is essential for the fertilization of mammalian oocytes. As an inhibitor of the oocyte-specific proteinase ovastacin, Fetuin-B maintains the fertilization ability of the oocyte. Consequently, Fetuin-B deficient mice are infertile. The oocytes of these animals show premature hardening of the zona pellucida (extracellular matrix surrounding the oocyte). Sperm are therefore unable to fertilize this oocyte. We measure Fetuin-B serum levels in humans and investigate the influence of Fetuin-B on oocyte fertilization ability in vitro and in vivo in a mouse model. Through structural analyses, we determine the domain of Fetuin-B that is essential for the inhibition of ovastacin. Thus, we are laying the foundation for potential therapies for patients with previously unexplained infertility, as well as for potential non-hormonal contraceptives.
Recently, we elucidated the exact interaction mechanism of fetuin-B and ovastacin using a 3-dimensional structural model.
Further Reading
Structure of mammalian plasma fetuin-B and its mechanism of selective metallopeptidase inhibition.
Cuppari A, Körschgen H, Fahrenkamp D, Schmitz C, Guevara T, Karmilin K, Kuske M, Olf M, Dietzel E, Yiallouros I, de Sanctis D, Goulas T, Weiskirchen R, Jahnen-Dechent W, Floehr J, Stoecker W, Jovine L, Gomis-Rüth.
FX.IUCrJ. 2019 Feb 28;6(Pt 2):317-330. doi: 10.1107/S2052252519001568. eCollection 2019 Mar 1. PMID: 30867929
Fetuin-B, a liver-derived plasma protein is essential for fertilization.
Dietzel E, Wessling J, Floehr J, Schäfer C, Ensslen S, Denecke B, Rösing B, Neulen J, Veitinger T, Spehr M, Tropartz T, Tolba R, Renné T, Egert A, Schorle H, Gottenbusch Y, Hildebrand A, Yiallouros I, Stöcker W, Weiskirchen R, Jahnen-Dechent W.
Dev Cell. 2013 Apr 15;25(1):106-12. doi: 10.1016/j.devcel.2013.03.001. Epub 2013 Apr 4. PMID: 23562279
Tissue Engineering
Materials used in contact with the human body are called biomaterials. Depending on the intended purpose, these materials should be inert, i.e. they should not cause any reactions of the surrounding tissue, or they should specifically direct the surrounding cells in a predetermined direction. We are investigating cell/biomaterial interactions to identify and optimize suitable combinations for bone replacement and cardiovascular tissue engineering. The biomaterials used are mainly polymers and ceramics. As main cell type we use adult multipotent mesenchymal stem cells, which differentiate into bone and smooth muscle cells. In addition, mesenchymal stem cells produce cytokines and growth factors that support wound healing and regeneration processes.
Mesenchymal stem cells
Mesenchymal stem cells (MSC) are adult, multipotent stem cells which can be isolated out of diverse tissue, such as bone marrow, umbilical cord Wharton´s Jelly, adipose tissue or placenta. They possess a self-renewal and a differentiation potential and can be differentiated in vitro e.g. towards bone, cartilage and adipose cells. Besides proliferation and differentiation, MSC release cytokines involved in diverse processes (angiogenesis, fibrosis, extracellular matrix remodelling) and they can direct immune responses. Thus, MSC are a promising cell type for Regenerative Medicine and Tissue Engineering.
Biomaterials
Classical biomaterials are ceramics, metals and polymers. There is a huge variety of biomaterials for medical funcions. Biocompatibility is very important for each biomaterial. This means that it has to comply with a specific function at a very specific area within or on the body, that it is non-toxic for adjacent cells and does not induce inflammation or rejection. So biocompatibility is related to in vivo situations, while in vitro, only cytocompatibility or cytotoxicity can be analyzed. Analyses are in accordance with the ISO 10993-5 „Prüfung auf in vitro-Zytotoxizität“.
The impact of biomaterials on a specific cell type can be induced or directed by diverse parameters, e.g. biomaterial chemistry, physical properties like the wettability, surface charge, and electrical conductivity. Besides, the surface topography has a strong impact on adherend cells.
Stem Cell/Biomaterial Interactions
There are two main goals in the focus of stem cell direction via biomaterial properties: stem cell proliferation or directed differentiation without additional differentiation factors. Besides iterative analyses of cell behaviour on single biomaterials, high-throughput analyses are developed in the last decade to investigate cell/biomaterial interactions. Nowadays, biomaterial arrays are suitable tools to analyze large sample sizes in parallel with the aim to identify cell/biomaterial combinations for tissue engineering applications.
Tissue Engineering
Tissue Engineering is an interdisciplinary research field combining the principles of natural and engineering science to develop biological substitutes to restore, maintain or improve tissue function (Langer und Vacanti, 1993). In most cases, a tissue engineering construct is composed of three main components, the biomaterial (scaffold), the cells and biochemical factors. It is of importance to understand the influence of a biomaterial on cell behaviour and to investigate their interaction in in vivo models.


![[Translate to Englisch:] [Translate to Englisch:]](/fileadmin/files/lehr-und-forschungsgebiete/zell-molekularbiologie-grenzflaechen/_processed_/4/b/csm_140403-UKA-001-01_d4dcd381c4.jpg)
![[Translate to Englisch:] [Translate to Englisch:]](/fileadmin/files/lehr-und-forschungsgebiete/zell-molekularbiologie-grenzflaechen/_processed_/e/d/csm_140403-UKA-013_1dadd05f6c.jpg)