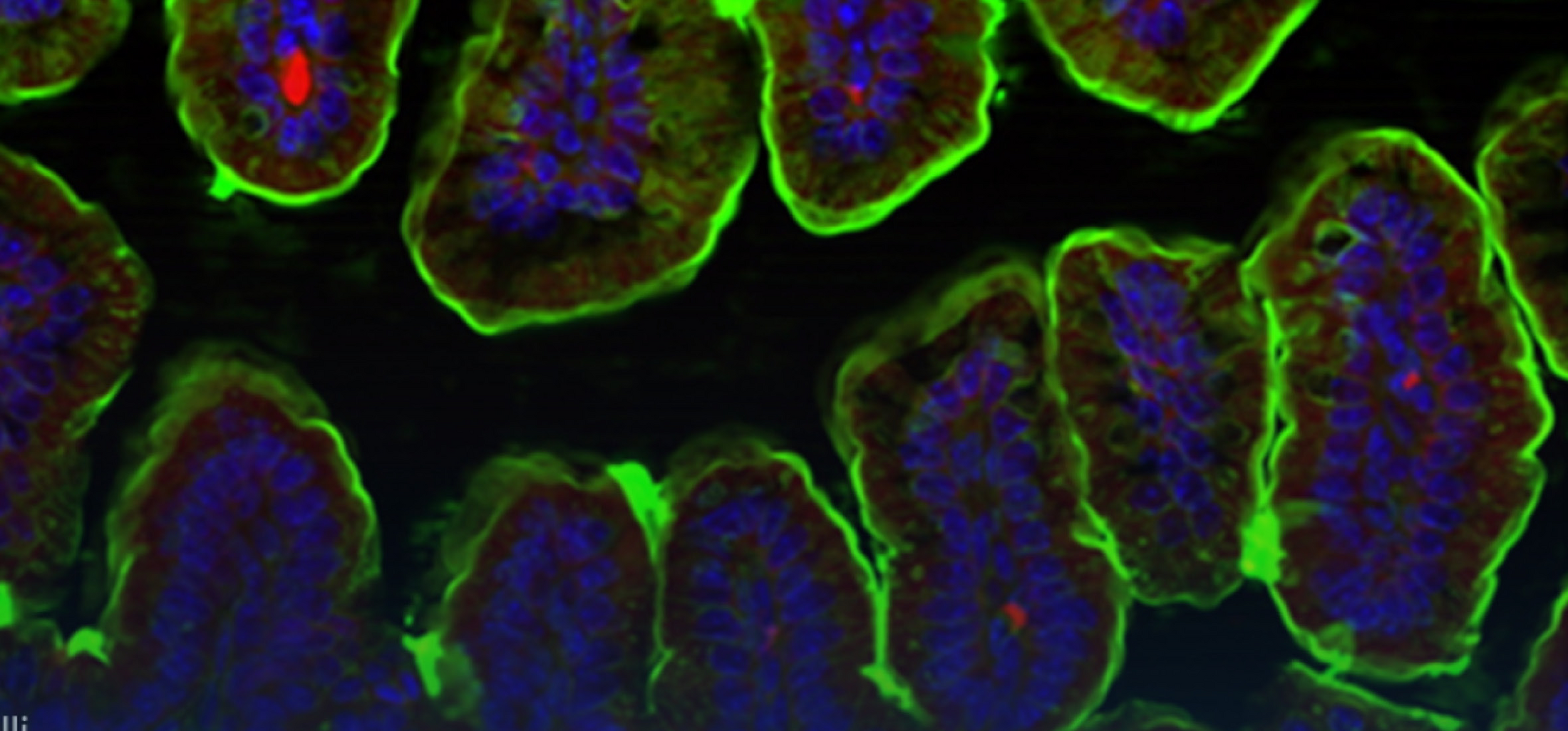
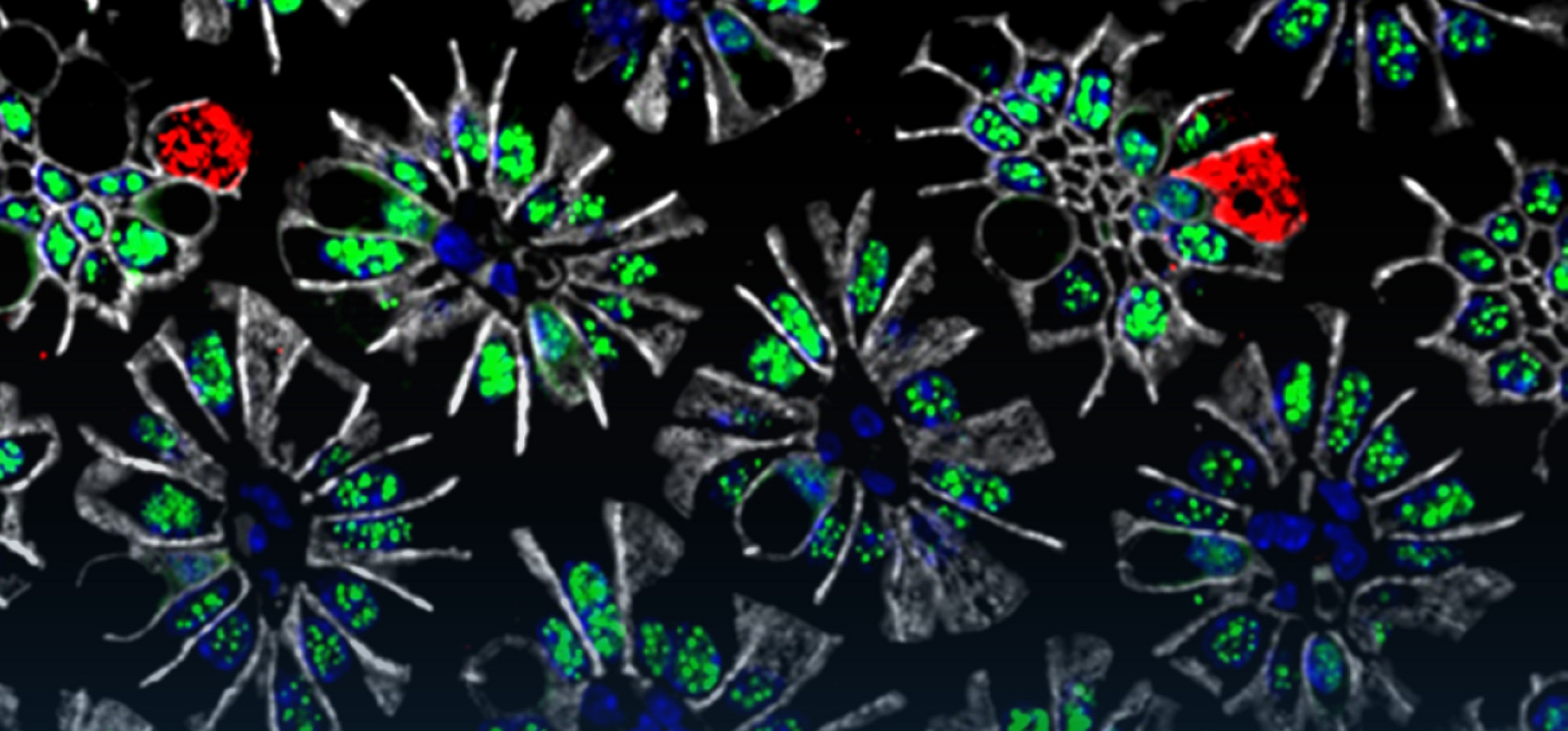
The intestinal immune system maintains a delicate equilibrium between the induction of immune responses to potential pathogens and the maintenance of tolerance to innocuous antigens, such as food and commensal bacteria. Disruption of this balance can lead to the development of deleterious inflammatory responses, such as food allergy or inflammatory bowel disease (IBD). Intestinal mononuclear phagocytes (MPs), comprising dendritic cells (DCs) and macrophages perform crucial roles in the control of intestinal immunity and tolerance; thus, they are attractive targets for immune therapies, including oral vaccines or induction of tolerance as treatment for food allergies and IBD. Our research studies the role DCs and macrophages play in the maintenance of tolerance and induction of intestinal immune responses, with particular emphasis on their effect on T cell activation and function.
Antigen presentation by intestinal macrophages in immunity and tolerance
Specialised subsets of MPs perform distinct functions. Intestinal DCs are uniquely able to migrate from the intestine to the mesenteric lymph nodes (MLNs) and initiate adaptive immune responses by priming naïve T cells. In contrast, macrophages are resident in the intestinal lamina propria (LP) and so are unlikely to play a major role in the priming of naïve cells. However, macrophages can perform crucial roles in the development of adaptive immune responses and intestinal tolerance. For instance, decreased IL-10 production by small intestinal macrophages results in reduced expansion of regulatory T cells and impaired oral tolerance. However, while intestinal macrophages express high levels of MHCII, their importance as antigen presenting cells in the development of CD4+ T cell responses has not been fully assessed. In this project we aim to characterise the molecular mechanisms that control MHCII expression on intestinal macrophages and their role as antigen presenting cells.
Liver dendritic cells in the induction of oral tolerance
Oral tolerance is the active process of local and systemic immune unresponsiveness to orally fed antigen. Failure of oral tolerance can lead to development of inflammatory bowel disease and food allergies. Recent reports have highlighted the crucial role played by intestinal dendritic cells (DCs) in the induction of Foxp3+ regulatory T cells (Tregs) and consequent tolerogenic responses in the intestine. However, it has long been known that the liver also plays a crucial role in the induction of oral tolerance. Despite this, relatively little is known about the cellular mechanisms responsible for the induction of tolerogenic responses to fed antigen in the liver, and how these two sites coordinate to establish oral tolerance. In this project we aim: 1) To characterise the liver lymph DCs for their phenotypic and functional properties with regards to induction of tolerogenic responses in the liver and gut 2) To characterise the functional and migratory properties of Foxp3+ Treg cells induced by intestinal and liver DCs in the context of oral tolerance 3) To examine how the tolerogenic function of DCs and the generation of peripheral Foxp3+ Tregs are altered in conditions of disrupted intestinal homeostasis.
Further reading
1. Cerovic V, Houston SA, Westlund J, et al. Lymph-borne CD8alpha+ dendritic cells are uniquely able to cross-prime CD8+ T cells with antigen acquired from intestinal epithelial cells. Mucosal Immunol. 2015;8(1):38-48.
2. Cerovic V, Bain CC, Mowat AM, Milling SWF. Intestinal macrophages and dendritic cells: what’s the difference? Trends Immunol. 2014;35(6):270-277.
3. Cerovic V, Houston SA, Scott CL, et al. Intestinal CD103(-) dendritic cells migrate in lymph and prime effector T cells. Mucosal Immunol. 2013;6(1):104-113.
Tolerogenic Function of Dendritic Cells in the Gut-Liver Axis
Gut and liver act as a unit to maintain immune tolerance in the steady-state, by inducing a population of regulatory T cells (Tregs). Intestinal dendritic cells (DCs) play a key role in Treg induction, however little is known about the tolerogenic potential of hepatic DC.
In this project we will characterize the role of hepatic and intestinal lymph DC in the induction of tolerogenic responses in the liver and gut. We compare functional and migratory properties of Tregs induced by hepatic and intestinal DCs. Moreover, we examine how the tolerogenic function of hepatic and intestinal DC is altered in conditions of disrupted intestinal homeostasis.
To experimentally address these Questions, we exploit a unique technique to capture migrating DC from mouse lymphatics that relies on adenectomy of the respective lymph nodes.
Funding:
This project is funded by Deutsche Forschungsgemeinschaft (DFG) in the context of Collaborative research Centre 1382 ‚Gut-Liver Axis – Functional Circuits and Therapeutic Targets‘ (https://www.crc1382.org/).