Functional characterization of heterologously expressed pain-related NaV mutants
This project is a subproject (P5) of the joint research consortium “Sodium channel‐related pain disorders: From molecular mechanisms towards personalized treatment” funded by the Aachener Interdisziplinäres Zentrum für Klinische Forschung (IZKF) of the Faculty of Medicine of RWTH Aachen University.
Chronic pain is a therapeutic challenge. Genetically encoded pain syndromes offer the chance to understand mechanisms of chronic pain on a genetic, cellular and molecular level and former studies could attribute multiple pain disorders to mutations in voltage-gated sodium channels (Dohrn et al., 2019). Human voltage-gated Na+ channels (hNaVs) are membrane proteins responsible for the rising phase of action potentials of excitable cells.
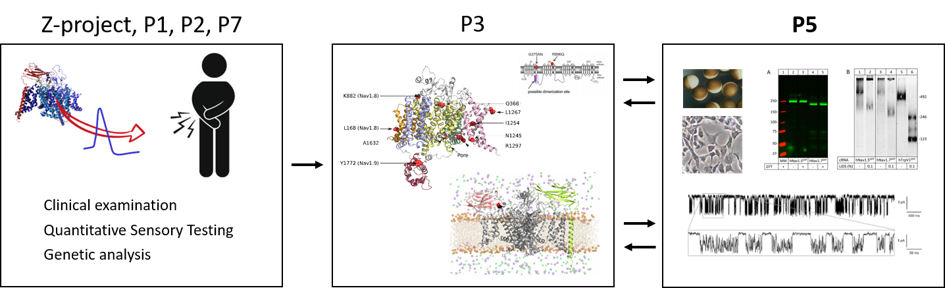
In one part of the subproject (principal investigator Priv.-Doz. Dr. med. Ralf Hausmann) we use an electrophysiological approach that allows us to address whether a particular pain-associated NaV variant affects NaV channel gating and provides novel insights into the biophysics of NaV channels in general and the mechanisms of how NaV mutants induce pain (Kaluza et al., 2018). In addition, single-channel recordings enable us to compare directly single-channel data with atomistic molecular-dynamics simulation data (P3).
In another part of the subproject (principal investigator Prof. Dr. med. Günther Schmalzing) we use biochemical techniques to address the expression level, stability, oligomerization and intra-cellular trafficking of recombinant hNaV1.7 and hNaV1.8 alone or together with auxiliary proteins including hβ subunits. These techniques allow addressing effects of point mutations that map to protein regions with known or unknown functions. Together with P1 and P3 we aim to analyze the oligomeric structure of hNaV1.7 and hNaV1.8 by a combination of structure-guided targeted mutagenesis, recombinant expression, NaV solubilization and native poly-acrylamide gel electrophoresis (PAGE).
Our dual biochemical-functional approach will allow us to address important questions such as whether a particular pain-associated NaV variant affects protein stability, dimerization or single-channel characteristics including coupled gating and activation or inactivation gating.
The figure below illustrates the integration of subproject P5 into the joint research project.
Dohrn et al., 2019. Neuropathische Schmerzsyndrome bei Ionenkanalerkrankungen. Der Internist 2019, 60:90-97.
Kaluza L, Meents JE, Hampl M, Rosseler C, Hautvast PAI, Detro-Dassen S, Hausmann R, Schmalzing G and Lampert A. Loss-of-function of Nav1.8/D1639N linked to human pain can be rescued by lidocaine. Pflugers Arch. 2018, 470:1787-1801.









