Our research group investigates the role of mechanical forces in various cell types and tissues of the human body, a field known as mechanobiology. A central focus is mechanotransduction, the process by which mechanical signals are converted into biochemical cellular responses. In particular, we study how inflammatory processes are regulated by mechanical influences, which are crucial for the healing of injured tissues and for the defense against pathogens.
A special emphasis of our work is the investigation of ADAM proteases and their regulators, which are considered key molecules in these processes. Through our basic research, we aim to create new approaches for the development of drugs, medical devices, and therapeutic strategies.
Our investigations cover several types of tissues, as mechanical forces play a role throughout the entire body. Currently, we focus on four main areas:
Vascular cells (particularly endothelial cells)
Lung cells (particularly epithelial cells of the alveoli)
Skin cells (particularly keratinocytes and fibroblasts)
Cells involved in bone formation and resorption (particularly osteoclasts and osteoblasts)
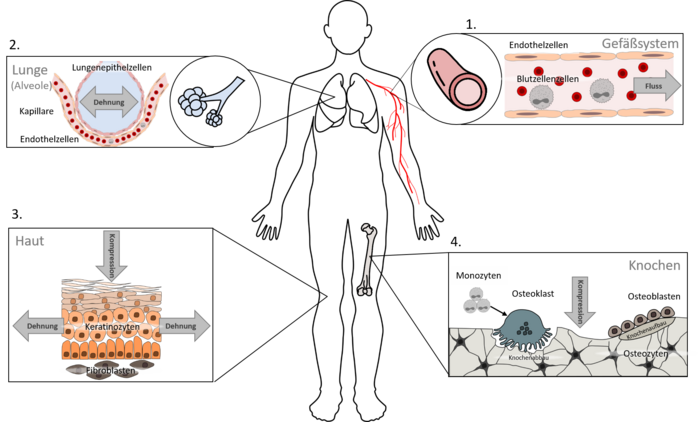
The investigation of mechanobiological questions naturally also requires the integration of mechanical forces into our cell culture experiments. For this purpose, we can rely on a broad interdisciplinary network, particularly through the graduate school ME3T (www.me3t.rwth-aachen.de). This allows us not only to culture cells under flow conditions, but also to stretch them (Merkel group, Institute of Biological Information Processing – Mechanobiology, Forschungszentrum Jülich GmbH), compress them (Fischer group, Department of Dental Prosthetics and Biomaterials), and inflict controlled mechanical injury.
Our collaborations also enable us to perform our investigations in complex skin models (Baron group, Department of Dermatology and Allergology), in ex vivo isolated perfused lungs and blood vessels (Martin group, Institute of Pharmacology and Toxicology), and in specialized animal models (Wolf group, Department of Orthodontics, and Ludwig group, Institute of Molecular Pharmacology).
Our previous results show that culturing endothelial cells under flow conditions has an anti-inflammatory and protective effect. For example, it influences the ADAM17 adaptor proteins iRhom1 and iRhom2 and promotes cell survival through increased ADAM15 expression. In addition, we found that the pro-inflammatory chemokine CX3CL1 is strongly reduced under flow conditions, which decreases the adhesion of immune cells to endothelial cells and thus limits the progression of inflammation in the surrounding tissue.
Recent findings demonstrate that ADAM10 and ADAM17 in lung epithelial cells and human umbilical vein endothelial cells (HUVECs) are stimulated through activation of the mechanosensitive ion channel Piezo1, revealing a new link between mechanical forces and ADAM protease–induced signaling cascades. Activation of ADAM proteases can lead to the release of a wide range of signaling molecules, which in turn can influence numerous inflammatory processes via autocrine, paracrine, or endocrine pathways. By understanding this signaling pathway, we aim to promote the development of new therapies for inflammatory diseases.
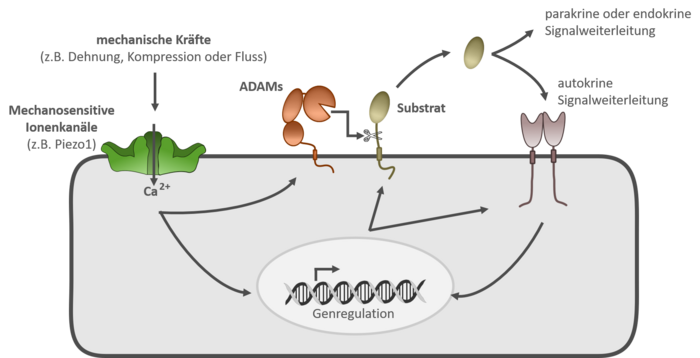
Methodologically, our work focuses primarily on cell biological approaches, which are consistently supported by molecular biological analyses.
| Cell culture | Cell Biological Methods | Molecular Biological Methods |
|
|
|
|
|
|
|
|
|
|
|
In addition to a flow cytometer (LSRFortessa, BD), our research group also has access to a live-cell imaging system (IncuCyte SX5, Sartorius), which enables monitoring of cells directly within the incubator. We are also happy to make these systems available to other research groups upon consultation. For further information, please feel free to contact Aaron Babendreyer (ababendreyerukaachende).
Publications
- Pabst, A., Brock N., Hardt S., Grannemann CCF., Kubiza H., Thiebes AL., Ludwig A., Babendreyer A. Mechanoresponsive ion channels Piezo1 and TRPV4 stimulate ADAM10 and ADAM17 with differential impact on endothelial migration and proliferation. Cell Commun Signal 2026;24:52. https://www.doi.org/10.1186/s12964-025-02633-x.
- Babendreyer, A.*, Kieselhorst J.*, Rinkens C.*, Lyashenko AM., Düsterhöft S., Jahr H., Craveiro RB., Wolf M., Ludwig A. Downregulation of the metalloproteinases ADAM10 or ADAM17 promotes osteoclast differentiation. Cell Commun Signal 2024;22:322. https://www.doi.org/10.1186/s12964-024-01690-y.
- Rojas-González DM.*, Babendreyer, A.*, Ludwig A., Mela P. Analysis of flow-induced transcriptional response and cell alignment of different sources of endothelial cells used in vascular tissue engineering. Sci Rep. 2023;13:14384. https://www.doi.org/10.1038/s41598-023-41247-6.
- Grannemann C., Pabst A., Honert A., Schieren J., Martin C., Hank S., Böll S., Bläsius K., Düsterhöft S., Jahr H., Merkel R., Leube R., Babendreyer, A.*, Ludwig A*. Mechanical activation of lung epithelial cells through the ion channel Piezo1 activates the metalloproteinases ADAM10 and ADAM17 and promotes growth factor and adhesion molecule release. Biomater Adv. 2023;152:213516. https://www.doi.org/10.1016/j.bioadv.2023.213516.
- Nußbaum SM., Krabbe J., Böll S., Babendreyer, A.*, Martin C.* Functional changes in long-term incubated rat precision-cut lung slices. Respir Res. 2022;23:261. https://www.doi.org/10.1186/s12931-022-02169-5.
- Giese AA.*,Babendreyer, A.*, Krappen P., Gross A., Strnad P., Düsterhöft S., Ludwig A. Inflammatory activation of surface molecule shedding by upregulation of the pseudoprotease iRhom2 in colon epithelial cells. Sci Rep. 2021;11:24230. https://www.doi.org/10.1038/s41598-021-03522-2.
- Babendreyer, A., Rojas-González DM., Giese AA., Fellendorf S., Düsterhöft S., Mela P., Ludwig A. Differential Induction of the ADAM17 Regulators iRhom1 and 2 in Endothelial Cells. Front Cardiovasc Med. 2020;7. https://www.doi.org/10.3389/fcvm.2020.610344.
- Babendreyer, A., Molls L., Simons IM., Dreymueller D., Biller K., Jahr H., Denecke B., Boon RA., Bette S., Schnakenberg U., Ludwig A. The metalloproteinase ADAM15 is upregulated by shear stress and promotes survival of endothelial cells. J Mol Cell Cardiol. 2019;134:51–61. https://www.doi.org/10.1016/j.yjmcc.2019.06.017.
- Babendreyer, A., Molls L., Dreymueller D., Uhlig S., Ludwig A. Shear Stress Counteracts Endothelial CX3CL1 Induction and Monocytic Cell Adhesion. Mediators Inflamm. 2017;2017:1515389. https://www.doi.org/10.1155/2017/1515389.



