Lungenpharmakologie
Measurement of lung function
Inflammatory and allergic lung diseases, such as ventilator-induced lung injury, pneumonia, asthma, chronic obstructive pulmonary disease or pulmonary hypertension are still difficult to treat. Frequently, disease models for these disorders are set up in vivo in laboratory animals (Flexivent) or ex vivo in isolated perfused lungs, but these models are complex and require a single animal for each experiment. In traditional airway pharmacology, tracheal rings, bronchial rings or isolated muscle tissue are used for in vitro measurements of smooth muscle contraction, but these models are also hampered by several issues, e.g. only large airways are accessible, contractions are either isometric or isotonic and relatively large volumes are required.
1. Applications of precision-cut lung slices (PCLS)
To circumvent the problems with in vivo studies and classical smooth muscle preparations we introduced the PCLS model, refined and extended it during the past 20 years.
PCLS are viable lung tissue slices of approximately 250 µm thickness with an intact microanatomy and a preserved cellular environment. This method lends itself to the study of airway responses such as airway contraction, and ciliary beat frequency, as well as to the study of vascular responses of both the pulmonary artery and the pulmonary vein. Depending on the species up to 50 PCLS (mouse, rat, guinea pig) or even more than 50 PCLS (pig, sheep, larger monkeys and human lungs) can be obtained from one animal. In so far, the PCLS method contributes to the 3R concept introduced by Russel and Burch and we were awarded by two animal welfare prices (Annual European Prize for the Substitution of Animal Experimentation 1997; animal welfare price of the Federal State of Schleswig-Holstein 1999).
The PCLS-Model was already used in the following pharmacological, physiological and pathophysiological studies in many different species, such as mouse, rat, guinea pig, baboon, macaque, marmoset, sheep, horse and human.
1.1. Inflammation in lung tissue
PCLS that are stimulated with cytokines, bacteria or rather bacterial components such as lipopolysaccharide (LPS) release pro-inflammatory cytokines. During our first cooperation period we have challenged PCLS with LPS and found a concentration dependent release of TNF, which was most prominent in PCLS from guinea pigs.
1.2. Physiological contraction of airways
PCLS are highly useful to study airways and vessels in the same experiment (figure 2). Depending on the species the physiological responses differ (see below).
PCLS provide an excellent model to establish concentration-response curves of endogenous and exogenous mediators. The effect of antagonists can be studied and their potency and IC50 can be compared. Schild-plots can be deduced from concentration-response curves to reveal the kind of antagonism (competitive, non-competetive) and possibly also binding-constants. The pharmacological characterization of knock-out or transgenic mice is also possible (51).
1.3. Contraction in airways of different size
The airway size plays a crucial role in chronic inflammatory lung diseases. For a long time the main focus of research was on the large airways, probably mainly due to their experimental accessibility. However, recently the important role of the small airways, i.e. airways < 2 mm diameter in humans, has been realized. Initially, the small airways were addressed by histology, but histology precludes functional analyses. The PCLS method, in contrast, allows to study airway responses along the entire tracheobronchial tree down to the small airways. Notably, depending on the mediator, the reactivity of small airways differs from that in large airways.
1.4. Neurally-induced bronchoconstriction in PCLS
Recently, we have established the electric field stimulation (EFS) of PCLS (53). The EFS-parameters frequency, pulse duration, current amplitude and train width were systematically adjusted to evoke reproducible airway contractions of neural origin (airway contractions are selectively inhibited by 10 mM magnesium). In the rat, we found a decreasing cholinergic innervation with decreasing airway size. The intrinsically-evoked cholinergic response in PCLS can be found in all species.
Across species, we found marked differences . Capsaicin (10 µM) caused bronchoconstriction in human and guinea pig lungs only, indicating excitatory non-adrenergic non-cholinergic responses (eNANC). In the most common experimental animals, i.e. in mice and rats, these responses differ considerably from those in humans.
1.5. Allergic response in PCLS
The early allergic response (EAR) is caused by the degranulation of sensitized mast cells. We also tested if allergens are capable to trigger bronchoconstriction in PCLS after passive sensitization with serum from actively sensitized subjects against ovalbumin or gras pollen. Again species differences were found: allergen application only elicited bronchoconstriction in PCLS from rats and humans but not from mice. The situation is somehow different in PCLS from actively sensitized animals.
We used the PCLS model to verify which mediators are involved in the EAR and found that leukotrienes and thromboxane mediate the EAR in human, while in rat the EAR solely depends on serotonin. ELISA measurements showed the release of serotonin and histamine in mouse, but this was not sufficient to induce-bronchoconstriction in mouse PCLS.
1.6. Measurement of ciliary beat frequency (CBF)
The mucociliary clearance is one important aspect of obstructive lung diseases. In this regard ciliary beating is extremely important to remove the mucus. The CBF can be quantified in PCLS, and stays stable for at least three days in human airways.
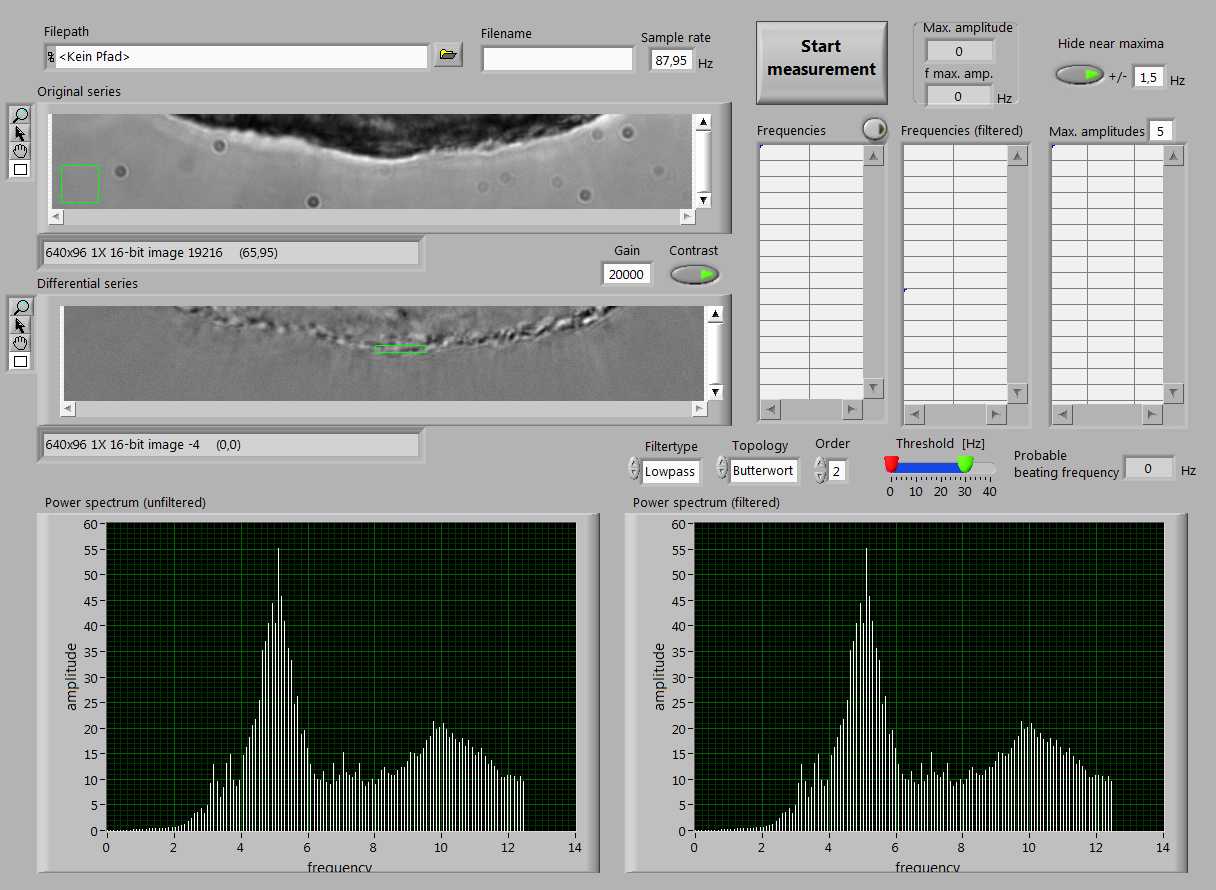
Recently, we have developed a new tool for analysing the CBF online. By continuous recording of images by a fast camera, coupled with online analysis of the intensity changes in the marked area, the CBF can be analysed in real-time (figure 4).
The CBF can be targeted pharmacologically, e.g. platelet activating factor decreases and endothelin-1 increases the CBF.
1.7. Reaction of pulmonary vessels in PCLS
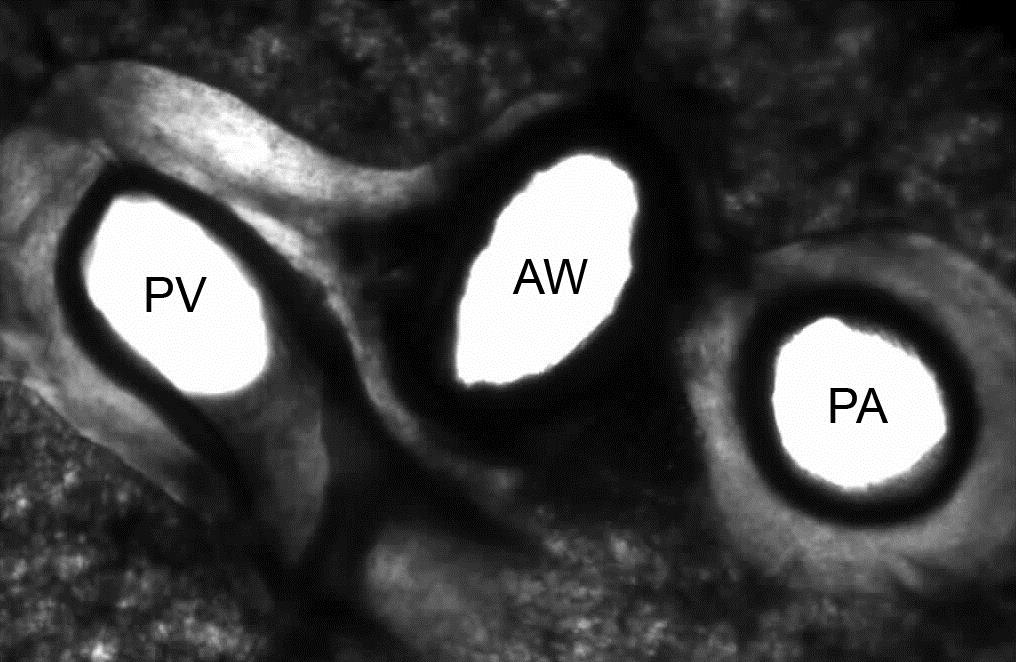
Besides the airways, also pulmonary vessels possess a smooth muscle layer and have an inherent tone. Elevated vascular tone crucially contributes to pulmonary hypertension, an important complication in COPD. Knowledge of the pulmonary vascular tone is also important for the treatment of cardiovascular diseases and right ventricular failure. Traditionally, only the pulmonary artery (PA) was analysed ex vivo in the organ bath experiments. However, clearly both PAs and pulmonary veins (PV) are important in pulmonary hypertension and global heart failure. Therefore, we have extensively studied the effect of contractile drugs on both pulmonary vessels in guinea pig PCLS. In brief, pulmonary veins responded stronger than arteries to α1-agonists (contraction) and β2-agonists (relaxation). Notably, inhibition of β2-adrenoceptors unmasked the α1-mimetic effect of norepinephrine and epinephrine in pulmonary veins. Vasopressin and angiotensin II contracted pulmonary veins via V1a and AT1 receptors, respectively, without affecting pulmonary arteries. Conclusively, PCLS provide a valuable tool to study the effects of mediators on pulmonary vessels (PA and PV) and to screen for agonists and antagonist acting on pulmonary vessels.
1.8. Multiphoton microscopy of PCLS
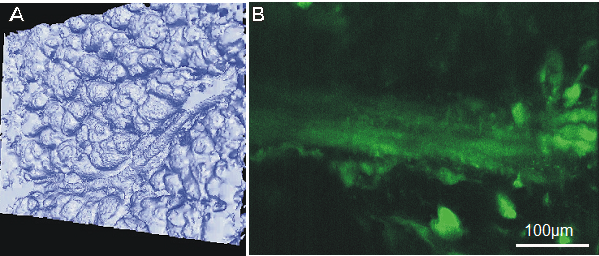
Pharmacological studies concerning airway or vessel reactivity require classical light microscopy. In contrast, cellular issues are not sufficiently addressed with light microscopy and are technically more demanding. We have recently established the two-photon microscopy of PCLS, in which a tissue penetration of 120 µm can be achieved at a local resolution of 250 nm. By this, a three-dimensional image of the lung with alveolar spaces can be constructed and observed over time (4th dimension) (figure 4A). In signal transduction processes, a cellular resolution is required and is achieved in PCLS. In figure 4B, for instance, PCLS were stained for calcium with Oregon green, and calcium fluxes became apparent in smooth muscle and epithelial cells.
1.9. Prevalidation of PCLS for acute respiratory toxicity
Due to REACH (Registration, Evaluation, Authorization and Restriction of Chemicals) and the 3Rs (refinement, reduction and replacement) principles, there is still a need of ex vivo models to test the acute toxicity of compounds. Therefore we studied PCLS as a probable model of acute toxicity in a multicenter study three independent laboratories (Fraunhofer ITEM, BASF SE, RWTH Aachen). The BfR provided important statistical support.
PCLS were prepared freshly and exposed to 5 concentrations of 20 industrial chemicals in serum-free cell culture medium under standard submerged culture conditions for 1 hour. After 23 hours post-incubation with serum-free cell culture medium, toxicity was assessed by released lactate dehydrogenase (LDH assay) and by mitochondrial activity (WST-1 assay). In addition, protein content and pro-inflammatory cytokine IL-1α were measured by BCA assay and ELISA, respectively. IC50 values were calculated after fitting oft he data by a sigmoid concentration-response model. For each endpoint test and procedure acceptance criteria are established and documented by SOPs.
By testing 20 chemicals, by analysing and fitting more than 900 concentration-response curves we obtained for all assay endpoints best intra- and inter-laboratory consistency for the data obtained by WST-1 and BCA assays. While WST-1 and LDH indicated toxic effects for majority of the substances, only some of the substances induced a significant increase in extracellular IL-1α.
These results show that the WST-1 endpoint is reliable for the use and PCLS were a powerful tool to measure acute respiratory toxicity.
1.10. Biomechanics can be determined by stretching of PCLS under defined conditions
The mechanical forces acting on lung parenchyma during (mechanical) ventilation and its (patho)physiological consequences are currently under intense scrutiny. Several in vivo and cell culture models have been developed to study the pulmonary responses to mechanical stretch. While providing extremely useful information, these models do also suffer from limitations in being either too complex for detailed mechanical or mechanistic studies, or in being devoid of the full complexity present in vivo (e.g., different cell types and interstitial matrix). Therefore we to develop a new model, based on the biaxial stretching of precision-cut lung slices (PCLS) together with the group of Prof. Dr. W. Wall at the TU Munich.
For biaxial stretching, single PCLS were mounted on a thin and flexible carrier membrane of polydimethylsiloxane (PDMS) in a bioreactor, and the membrane was stretched by applying varying pressures under static conditions. Distension of the membrane-PCLS construct was modeled via finite element simulation. Alternatively uniaxial tensile tests allowed the stretching of living precision-cut rat lung slices. Image registration is used to compute the displacement field at the surface of the sample. The constitutive model consists of a hyperelastic potential split into volumetric and isochoric contributions and a viscous contribution. This allows for the description of the experimentally observed hysteresis loop. The aim of these studies is to create a mathematical model for different parts of the lung. In this context the role of fibers, ground matrix and cells were studied.
2. Applications of isolated perfused lungs (IPL)
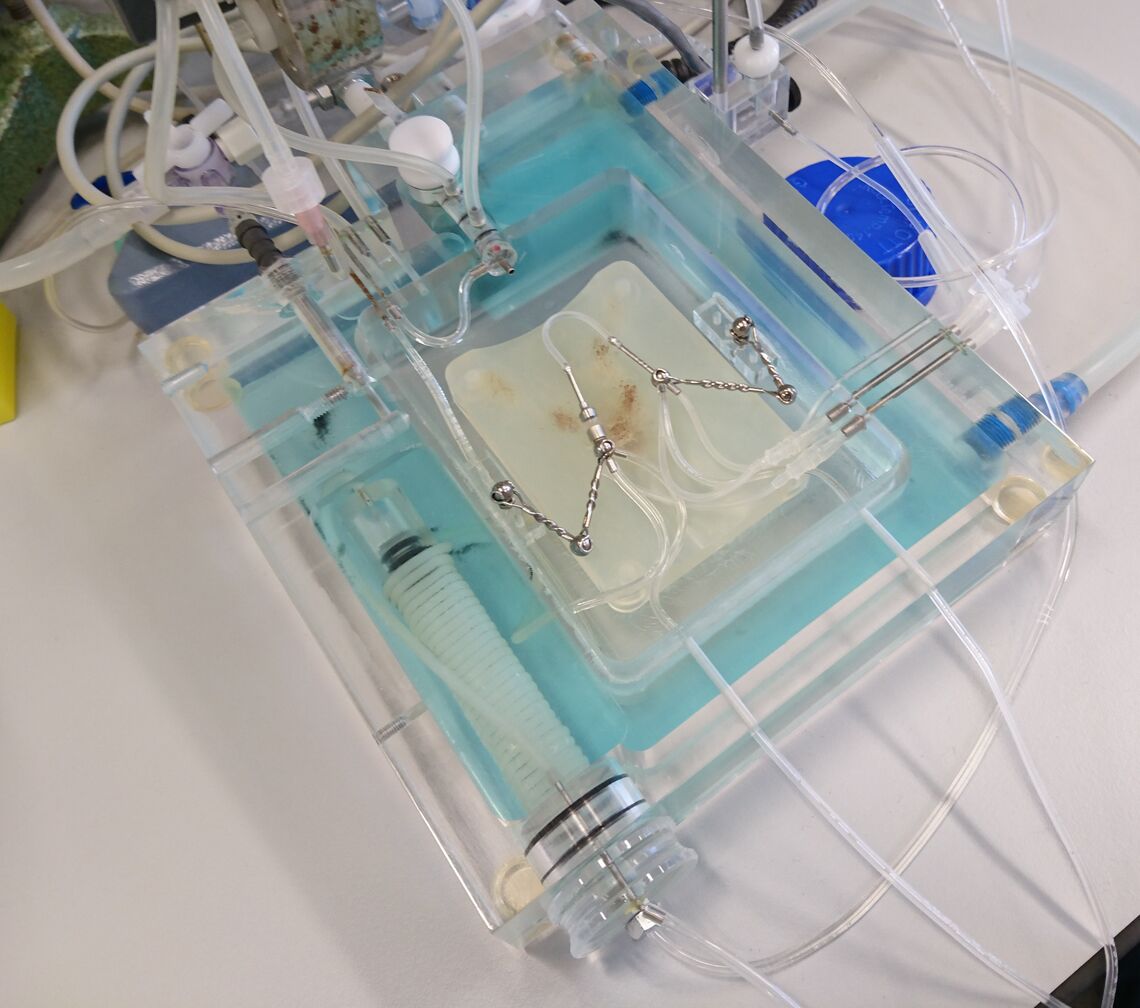
The isolated perfused lung model offers numerous possibilities to investigate the lung and pulmonary circulation. The lung is ventilated via tracheal cannula and negative pressure ventilation in a closed chamber, while the pulmonary circulation is perfused via the pulmonary artery. Pulmonary artery pressure, pulmonary venous pressure, pre- and postcapillary resistance can be determined, as well as parameters of lung mechanics. Investigations include effects exerted by various substances added to the perfusing buffer, instilled via trachea or effects of different inspiratory gas mixtures, e.g. hypoxic gas. The institute of pharmacology and toxicology has three different IPL settings for mice, rats and guinea pigs, figure 5 shows a murine IPL.
2.1. EphrinB2 and ephB4
The arterial and venous systems are distinct entities, different up to their smallest compartments. The receptor tyrosine kinase ephB4 is expressed solely in venous endothelial cells and its ligand ephrinB2 is specific for arterial endothelial cells, smooth muscle cells and their surrounding pericytes. Thus, those two markers define the border between arterial and venous systems and determine the arterial or venous identity of vessels. EphrinB2 and ephB4 are not only markers of vessel identity, but crucial regulators of endothelial cell migration and proliferation.
In this project the role of ephrinB2 and ephB4 in the pulmonary circulation and especially acute lung injury is investigated. Our models include the isolated perfused mouse lung, as well as murine and human precision cut lung slices.
2.2. Acute hypoxia – Hypoxic pulmonary vasoconstriction (HPV)
HPV represents an elaborate network of mechanisms leading to low arterial partial pressure of oxygen triggered pulmonary vasoconstriction to match local perfusion and ventilation. It includes modulation of K+-channels, alteration of mitochondrial reactive oxygen species production, modulation of hypoxia-inducible factor, cyclooxygenase, and lipoxygenase, resulting in vasoconstriction.
The effects of acute hypoxia and repetitive hypoxic maneuvers in terms of HPV on the pulmonary circulation are studied. Since hypoxia is a known trigger for ephrinB2 expression, the connection of ephrinB2 and ephb4 with hypoxia will be examined.
2.3. Effects of different hydroxyethyl starches and gelatin on the pulmonary circulation
Plasma expanders are frequently used for patients during surgery and in intensive care. They contain substances to increase the oncotic pressure like hydroxyethyl starch (HES) or polysuccinated gelatin. However, both HES and gelatin exert also adverse effects, e.g. acute kidney injury or dilutional coagulopathy. In vitro and in vivo, anti-oxidative and anti-inflammatory properties have been attributed to HES and pro-inflammatory responses up to anaphylactic shock to gelatin.
In isolated perfused lungs we are able to study the lung without interference of different factors such as blood, the nervous system or other organs. Influence on physiological parameters including tidal volume, airway resistance and pulmonary artery perfusion pressure can be determined, as well as lung cytokine production and oedema formation. Thus, the effects of colloid solutions on the lung can be thoroughly examined.
These projects were organized and performed by Dr. med. Julia Krabbe.
3. Measurements of lung mechanics (FlexiVent-ventilation setup)
The flexiVent (Scireq) is widely regarded as the gold standard for in vivo lung function measurements especially of LPS-treated animals or asthmatic mice. It offers the possibility to study traditional resistance and compliance, but also helps to investigate mechanical properties of conducting airways, terminal airways and parenchyma. Additionally, airway hyperresponsiveness can be assessed in response to aerosol challenge, e.g. acetylcholine or metacholine. During measurements of lung mechanics mice are tracheotomized and physiological data, such as rectal temperature, heart rate and ECG, are recorded to estimate general conditions during the experiments. Fig. 6 shows the FlexiVent setup including data acquisition and control units and monitoring.
3.1. Lung mechanics in asthmatic mice
Allergic diseases represent significant global health problems with dramatically increasing morbidity during the last decades. Allergies can affect people of all ages, however the prevalence in childhood is up to 39 %. Allergic asthma is characterized by airway inflammation, recurrent bronchospasms, airway hyperresponsiveness (AHR) and mucus hypersecretion. The asthmatic response develops as a crosstalk between innate cells and the adaptive immune system with TH2 cells becoming important mediators of allergic inflammation. Disease activity depends on numerous complex mechanisms involving T lymphocytes. Therefore, we could demonstrate that T cell dysregulation, e.g. by regulation of transcription factors (cAMP responsive element modulator (CREM)) can clearly influence asthmatic response and lung mechanics in Ovalbumin-induced asthma.
In our current research project, we focuse on the acid sphingomyelinase (ASM), an that belongs to the sphingomyelinase (SMase) family and is responsible for catalyzing the breakdown of sphingomyelin to ceramide and phosphorylcholine in the ceramide generation. Its deficency causes lipid storage diseases, Niemann-Pick-disease (NPD) type A (neuronopathic) and B (non-neuronopathic). Previous studies demonstrated an association of ASM deficiency or inhibition and better outcome in cystic fibrosis. We investigate the role of ASM deficiency in a murine model of allergic asthma.
This project is funded by the DFG (Deutsche Forschungsgesellschaft).
3.2. Lung mechanics in acute lung injury
Acute lung injury (ALI) is a common serious disease in critically ill patients involving high mortality and morbidity. Mechanical ventilation is commonly used as main therapeutical strategy although changes of lung mechanics during different stages of ALI are not clearly defined. Additionally, patients with immunodeficiency (e.g. SCID with T and B cell deficiency) are supposed to be highly susceptible for serious ALI, but differences in lung mechanics compared to immunocompetent patients and therefore the role of T lymphocytes on lung function changes in ALI are only rarely understood. Further studies of our group showed that a dysregulation of T lymphocytes could trigger inflammation.
In our recent project, we now investigated the role of non-dysregulated T lymphocytes in different phases of disease. Therefore, LPS is instilled intratracheally on day 0 and we measure lung mechanics, inflammatory markers in bronchoalveolar lavage and lung tissue on day 1, 4 and 10 after ALI induction.
This project is funded by the DLS (Deutsche Lungenstiftung).
These projects were organized and performed by Dr. med. Eva Verjans.