Wissenschaftliche Arbeitsgruppen und Forschungsfelder
AG Boor: Our research focuses on improved diagnostic, understanding of progression and treatment of chronic kidney disease (CKD), the common end-point of virtually all renal diseases, and on cardiovascular consequences of CKD. From methodological perspective, our main focus and expertise is in using, developing and refining animal models of kidney diseases, invasive and non-invasive in-vivo imaging, full spectrum nephropathological analyses (including electron microscopy) and artificial intelligence (particularly deep learning). (www.LaBooratory.ukaachen.de)
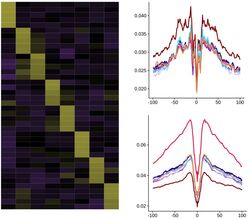
AG Kuppe: Every cell, the basic unit of life, has its own story to tell! Additionally, the surroundings of the cells, its’ neighborhood and the organ the cells reside in, impact cellular functions in healthy and disease. In our research, we are particularly interested in the molecular signals that define cellular interactions in different human organs and the analysis of temporal changes along disease specific trajectories. The lab focuses on the application of novel single-cell and spatial multi-omic approaches to decode disease trajectories thus defining novel cellular disease mechanisms by including temporal and spatial information. (www.kuppelab.com)
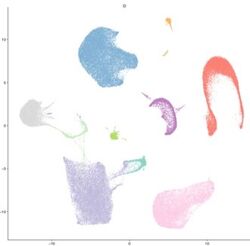
AG Kramann: We are studying renal and cardiac fibrosis and atherosclerosis and aim to identify novel therapeutic targets via single cell genomic technologies. We use genetic fate tracing, transgenic animal models, in vitro and in vivo CRISPR/Cas9 gene editing and organoids to preclinically validate targets and develop new therapeutic strategies (Institut für Experimentelle Innere Medizin und Systembiologie)
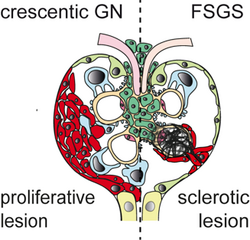
AG Moeller: We are investigating glomerular diseases in the kidney to develop novel therapeutic concepts. We also study how the kidney filter might work and why glomerular disease leads to proteinuria. A third focus are similar injury-induced transcriptional programs which play different roles in the glomerulus and the proximal tubule in acute kidney injury. For this we employ a broad spectrum of available methods such as transgenic animal models, isolated perfused kidneys, telemetric blood pressure measurements, high-end microscopy and single-cell transcriptomes.
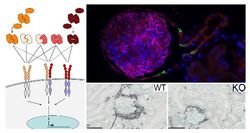
AG Ostendorf/Floege: In the context of diagnosis and therapy of glomerular and tubulointerstitial kidney diseases, we focus on the research of central, pathophysiologically relevant mediators and their receptors in preclinical studies. In the recent past, we identified pro-fibrotic, pro-angiogenic, blood pressure-regulating and anti-proliferative molecules and systems that play a central role in the progression of kidney diseases and whose manipulation in preclinical medicine has shown new therapeutic potential. Using a broad spectrum of experimental models of renal inflammation and kidney damage, (conditional) transgenic animals, up-to date molecular and histological techniques, we currently focusing on studying a) the role of platelet-derived growth factor C (PDGF-C) and its receptor, b) the renal serotonin/serotonin receptor system in acute and progressive renal diseases and c) consequences of renal cell compartmentalization defects.
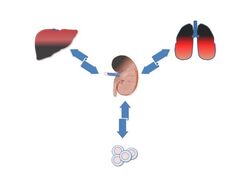
AG Raffetseder/Rauen: The goal of our research is to understand the molecular and cellular mechanisms of kidney disease and inflammation. We focus on the role of the highly conserved Y-box binding protein (YB-1) and its receptor Notch-3. In addition to organ-specific processes driven by YB-1 and Notch-3, we are also interested in the interaction between the kidney and other organs and the influence of the YB-1:Notch-3 axis on these organ cross-talks. Our methodological spectrum covers diverse kidney inflammation/damage models, transgenic animal models, and a broad spectrum of cutting-edge molecular techniques as well as histological and molecular tools.



Neue Methode der schnellen Diagnose von Lupusnephritis bei SLE-Patienten (EFRE-0400348)
Der systemische Lupus erythematodes (SLE) ist eine Autoimmun-erkrankung und betrifft weltweit mehrere Millionen Menschen. Die damit assoziierte Lupusnephritis stellt dabei das Hauptrisiko für Morbidität und Mortalität dar und erfordert spezifische Therapie. Ziel dieses Projektes ist die Entwicklung einer schnellen, zuverlässigen, kostengünstigen und im Klinik- und Praxis-Alltag unkompliziert einsetzbaren Lupusnephritis-Diagnosetechnik.
















