Research projects and techniques
Only a fraction of genetic epilepsies is explained by single genetic variants. The larger share of genetic epilepsies is based on a complex genetic architecture that implies the influence of numerous genetic and potentially epigenetic factors. Even monogenetic epilepsy syndromes often feature a remarkable interindividual phenotypic heterogeneity that suggests an interplay of further genetic factors. Drug-resistance i.e., the failure to achieve seizure freedom despite adequate medication, presents another of the great challenges in epileptology.
The aim of our work group is contributing to achieve a better understanding of the genetic architecture of epilepsies and to investigate genetic biomarkers that could help improve diagnostics and genetic counseling in the epilepsy field. We also seek to determine genetic predictors for individual drug response to anti-seizure medication and post-surgical outcome to pave the way to personalized therapeutic pathways.


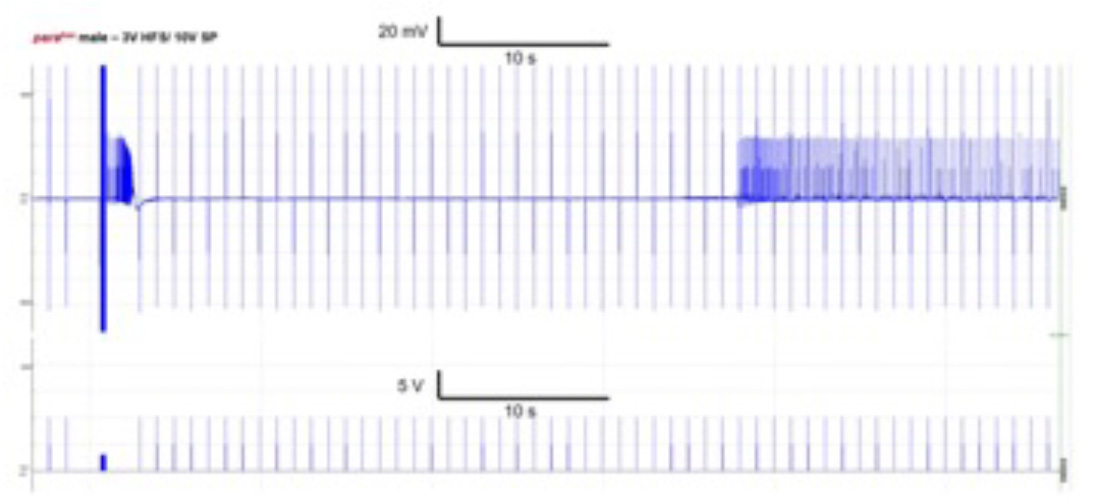
Drosophila melanogaster is an established epilepsy model with multiple epileptic phenotypes. Besides well-known epilepsy fly lines, such as parabss1 and eas, we use different methods to generate transgenic flies (GAL4/UAS, CRIMIC) that either express altered fly genes or humanized genes. We focus on synaptic epilepsy genes, such as AP2M1, STXBP1, and STX1B that have a high degree of orthology with fly genes. We use behavioral experiments, Calcium-imaging, and diferent electrostimulation techniques to elicit specific epileptic phenotypes. We also use various anti-seizure medications to study their effect on the epileptic phenotypes in flies.
Electroencephalography (EEG) is one of the mainstays for the diagnostic and assessment of people with epilepsy and other seizure-like episodes. Long-term-EEG recordings via video-EEG-monitoring and invasive recording methods, such as stereo EEG (sEEG) and intraoperative electrocorticography provide indispensable insights to tailor surgical interventions for drug-resistant epilepsy.
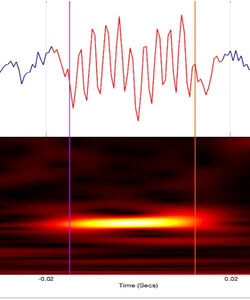
The aim of our work group is the investigation of EEG signals as biomarkers for diagnostics and outcome prediction. We employ biosignals from multiple EEG modalities: video-EEG-Monitoring, routine EEG, 128-channel-EEG, sEEG and intraoperative ECoG. The analyses comprise the evaluation of physiological signals, the assessment of high frequency oscillations (HFOs), and the EEG post-processing based on microstate analyses.
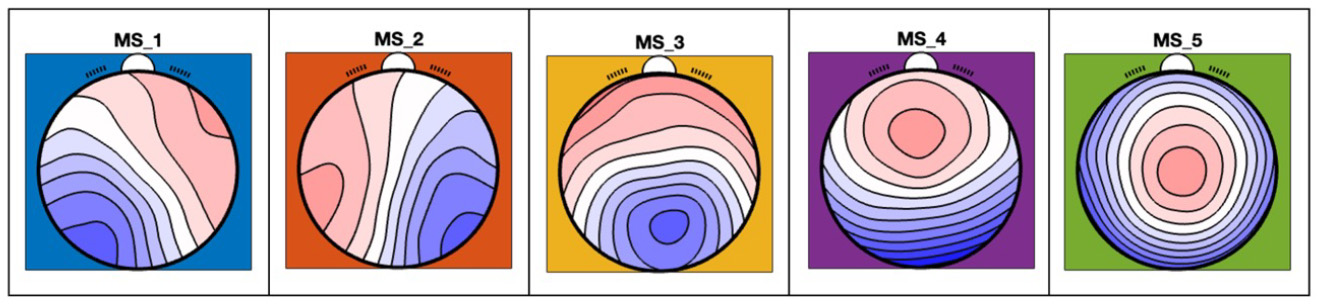
In direct cooperation with the Clinic for Psychiatry, Psychotherapy, and Psychosomatic Medicine (work group L. Wagels) we currently investigate imaging and neuropsychological biomarkers for aggressive behavior in people with epilepsy. The focus of this project is on aggressive behavior as an adverse reaction to anti-seizure medications (ASM). We employ various paradigms to incite and measure aggressive behavior und assess brain functional characteristics. This research could help develop clinical predictors for ASM tolerability and thus improve the safety of the therapeutic management.















